How Study Participants can Access Their Activities and Forms
Table of Contents:
- Participant Experience
- Accessing forms
- Completing or Revoking Consent
- Accessing Past Due or Completed Forms
- Issues With Form(s) Missing
- Getting Erroneous Notifications
- Switching to another study
- ePRO Forms Due for Completion Questions/FAQs
Participant Experience Walkthrough
Home Screen and Form Access
Once you are signed in, depending on the platform used (Android, iOS, or Web) the app will open to a home screen displaying content that varies by study. In any case, the forms due for completion will be readily accessible.
If no forms are showing as due and you think there should be, please contact your study or site Coordinator to verify something should be available.
NOTE: The TrialKit help desk will not be able to support questions related to specific studies, but can help with issues related to the website or app.
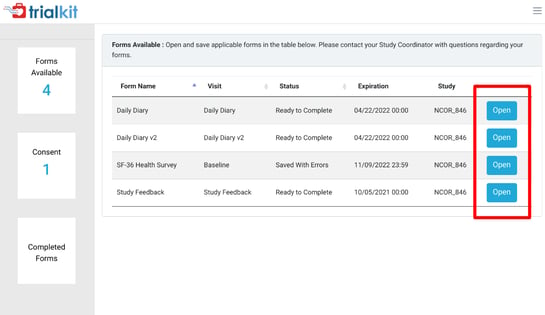
Web Browser:
Android:
On Android the forms will be available on the main home screen. Other available tools are dependent on the study.

iOS:
On iOS, tap the "Show My Forms" button. Other available tools available on the home screen are dependent on the study.

Consenting To a Study
If the study requires consent, you will see a prompt to complete the Consent form prior to accessing any other forms.
Mobile App:
To provide consent, tap the Consent option in the menu.

Once the Consent form is completed, you will receive an email verification with the attached documents.
If the study is set up to allow you to revoke consent from the app, this can be done via the icon indicated below, found at the top of the Consent form as shown below.

Accessing Expired or Previously Completed Forms
Named:
1. "My Expired Forms" on the IOS and Andriod App or "Expired Forms" on Web Patient Portal
2. "My Completed Forms" on the IOS and Andriod App or "Completed Forms" on Web Patient Portal
Some studies may allow you to access forms that were missed or forms that were completed but are no longer available on your primary list of due forms.
Those can be accessed within the menu on both the web browser and mobile app.
Web Browser:

Issues With Forms Missing
If you have signed in to complete a form or received a notification that a form is due, but are unable to see or access the form, there is likely a problem with the configuration. In this case, you would need to contact the Study Coordinator or the study contact provided to you for support, so they can escalate it to the Study Administrator.
For Help with the app and technical issues, tap the help button at the bottom right corner from the web browser or mobile app.
**Please note the TrialKit support desk is unable to answer any study-related questions, including the intended behavior of a given study within the TrialKit application. It is best to direct questions to your study Coordinator or the contact provided by the Sponsor when you were initially registered for the study. **
Switching to another study
If you are part of two or more studies being conducted on the TrialKit platform, it will be necessary to switch between those studies within the app to see the applicable content.
For example, you might have received a notification that something is due to complete, but when you sign in, there is nothing available to complete. If this is the case, and you know you have Participated in more than one study on TrialKit in the past, it's likely that you need to toggle over to the newer study.
To switch between Sponsors and studies, use the menu options highlighted below:
Web browser:

iOS:

Android:


ePRO Forms Due for Completion Questions/FAQs
1. I received a notification, but nothing is available when I sign in.
There are two possibilities in this scenario.
-
The form or action item has expired based on how it’s been configured by the Study Administrator.
-
Some studies may utilize an external notification method (e.g. email) which may not necessarily follow the form workflow designed in TrialKit.
What to do: Contact your Study Coordinator to inquire about a form that may have been missed due to expiration. If the system is incorrectly sending notifications, they should escalate that issue to the Study Administrator for correction.
2. The notification I received does not tell me anything.
Notifications are configured by the Study Administrator in the form builder and notification configuration.
What to do: Please escalate this to your Study Coordinator so it can be corrected.
3. I’m supposed to fill a form out but don’t see anything on my list.
This means nothing is currently open to fill out. The form could have expired already or there is nothing you need to fill out at this time.
What to do: For questions about activities to complete, please contact your Study Coordinator.
4. Why Do I still get notifications for a study that I am no longer part of?
This means the Study Coordinator has not correctly removed your participation status. You can contact them directly to get that done.
Avoid unsubscribing from email notifications if possible. If you do unsubscribe, you will not get pertinent study notifications in the future (if applicable to the study).
If you believe you are receiving notifications erroneously, please contact the Study Coordinator or person that helped you register for the study.
5. Do I need to download the mobile app
No. TrialKit is also available via web browsers at this URL: participant.trialkit.com
